Nexium Control overview

The UK’s No 1 for 24-hour heartburn protection1
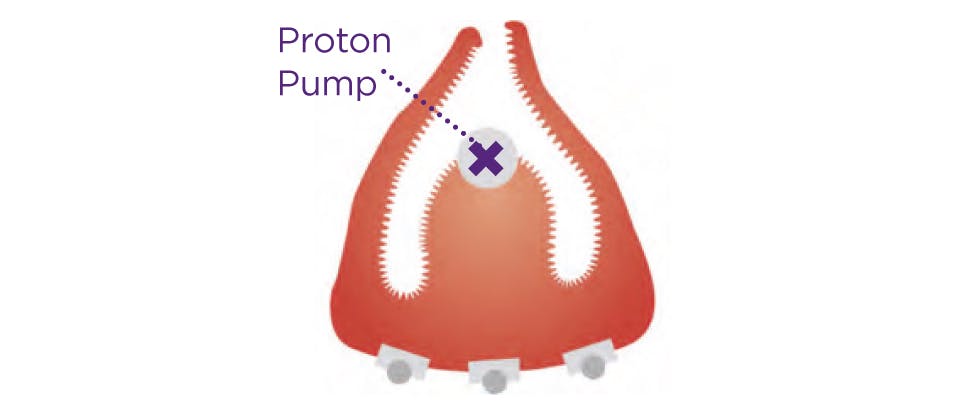
Unlike antacids and alginates that treat the symptoms of heartburn, Nexium Control tackles the cause by targeting acid directly at the source providing long-lasting protection.
For news, updates and access to more resources and webinars please register now to be on our mailing list.
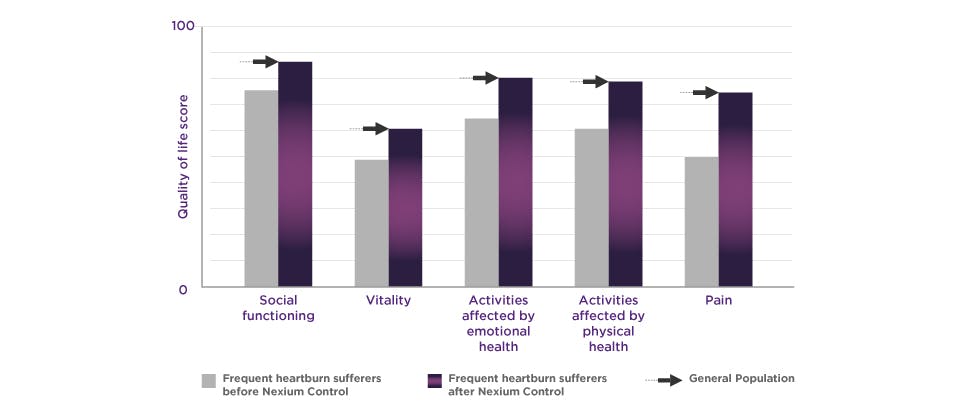
Nexium Control – Clinically proven protection for frequent heartburn

Long lasting protection
With just one pill a day Nexium Control provides up to 24 hour protection for frequent heartburn sufferers.

Clinically proven
8 out of 10 people reported first resolution within the first week of treatment with Nexium Control.

Nighttime relief
4 out of 5 nights were heartburn free for patients using Nexium Control.
Nexium Control – Your first line recommendation for frequent heartburn
Find out more about frequent heartburn
Explore videos, product guides and other resources to support you and your team when recommending Nexium Control for frequent heartburn.
Recommend Nexium Control for your patients with frequent heartburn
Nexium Control 28 tablet pack
For patients with recurrent frequent heartburn who have used esomeprazole successfully and seek the value and convenience of a larger pack.
Nexium Control 20mg Gastro-Resistant Tablets (Esomeprazole)
Nexium Control 7 tablet pack is an ideal trial pack with one-week supply. Nexium Control 14 tablet pack offers the same protection and offers a 2-week treatment. 2-3 consecutive days treatment may be needed to achieve improvement of symptoms. The duration of treatment is up to 2 weeks. Once complete relief of symptoms has occurred, treatment should be discontinued.
If symptoms worsen or if no symptom relief is obtained within 2 weeks of continuous treatment, the patient should be instructed to consult a doctor.
Nexium Control 20mg Gastro-Resistant Capsules (Esomeprazole)
Easy-to swallow mini capsules – 53% smaller than tablet* in a portable bottle. Complete 14-day treatment. 2-3 consecutive days treatment may be needed to achieve improvement of symptoms. The duration of treatment is up to 2 weeks. Once complete relief of symptoms has occurred, treatment should be discontinued.
If symptoms worsen or if no symptom relief is obtained within 2 weeks of continuous treatment, the patient should be instructed to consult a doctor.
*based on volume
Nexium Control – the UK’s No 1 for 24-hour heartburn protection.1
Heartburn
Find out more about frequent heartburn including a profile of a patient who may suffer from the condition.

Webinars & Events
Register now to access a range of webinars and education modules online.
Educational resources
Access educational resources to support your team’s understanding of heartburn and its management.