Voltaren Emulgel 2%

Voltaren Emulgel 2% − eases joint pain and inflammation all day long when applied twice daily1
Provides up to 12 hours of relief from joint pain through an anti-inflammatory action.1
Contains diclofenac, a potent non-steroidal anti-inflammatory drug (NSAID) with effective analgesic, anti-inflammatory and antipyretic properties.2
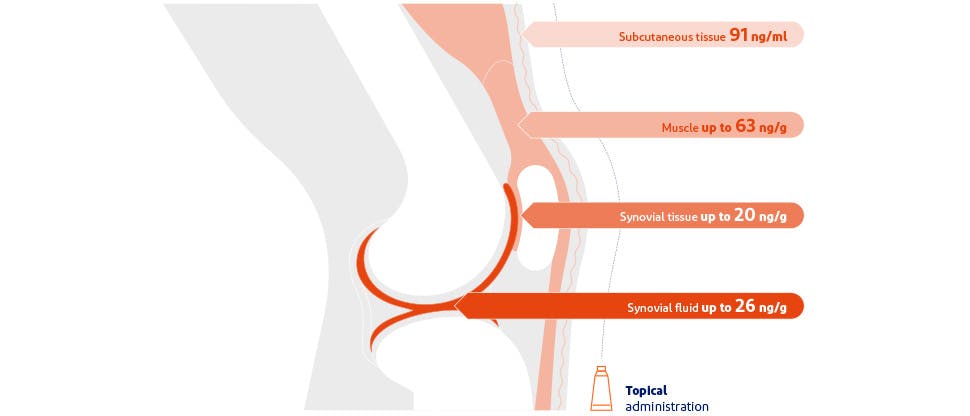
Diclofenac gets deep into the joint area3
After topical administration, diclofenac is distributed to the subcutaneous tissues, muscle, synovial tissues and synovial fluid, the latter tissues typically being most likely to be driving joint pain.3 Its chemical properties support retention within the joint where it is needed, rather than the blood stream.3,4
Voltaren Emulgel 2% is specially formulated to help the anti-inflammatory ingredient get through the skin.5,6 It contains a permeation enhancer for increased delivery of diclofenac through the skin.6*
The deep penetration of Voltaren Emulgel 2% makes it suitable for the treatment of joint pain related to trauma of the tendons, ligaments, muscles and other joint tissues as well as localised soft-tissue rheumatism.5
*Voltaren Emulgel 2% compared with Voltaren (1.16%) Emulgel (in vitro study) when measured over 24 hours.

Provides up to 12 hours of relief from joint pain1
Voltaren Emulgel 2% has proven clinical efficacy: in a study of acute ankle sprain, patients experienced a 65% reduction in pain from baseline after 4 days, with additional benefits in terms of reductions in swelling vs placebo.1
Voltaren Emulgel 2% provides all-day joint-pain relief, meaning convenient twice-daily application is all that is needed.1,5

Low systemic absorption, for a lower risk of systemic side effects3,5,8
Voltaren Emulgel 2% has a low systemic exposure3,5,9 meaning a reduced risk of systemic side effects and drug–drug interactions.3,5
That includes a reduced systemic side-effect risk vs oral NSAIDs (e.g. gastrointestinal events).10,11*
*There is an increased risk of mild dermatological side effects (e.g. skin irritation) with topical diclofenac, but this is non-serious and reversible.
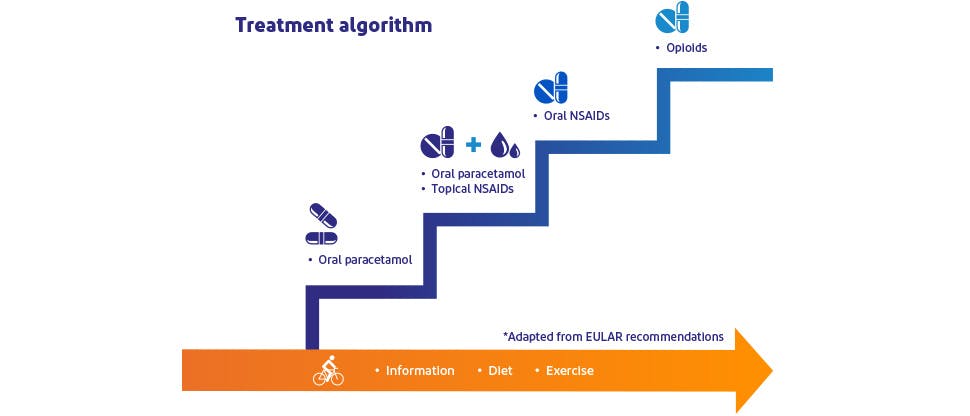
Providing additional pain-relief alongside paracetamol12–14*
Clinical guidelines support the use of paracetamol for chronic osteoarthritis pain of the knee,12,15 hand12 and hip,15 with the option of adding topical NSAIDs for further pain relief,12,15 including for the treatment of short-term pain flares that often accompany chronic osteoarthritis pain.15 However, more-recent guidelines do not recommend the use of paracetamol (alone or in combination with topical NSAIDs) for the treatment of osteoarthritis.16
*Current Osteoarthritis Research Society International (OARSI) guidelines do not recommend treating osteoarthritis with paracetamol.
†Current OARSI guidelines do not recommend treating osteoarthritis with oral or transdermal opioids.

Nora needs to regain mobility by easing joint stiffness
Nora wakes up with joint pain and stiffness each morning and has difficulty climbing stairs but does not want this to control her life or her choices.
She already receives multiple oral medications and is concerned that adding more will increase her risk of side effects.
Nora tries to stay active by gardening and going for walks, but this can sometimes be a struggle.
She wants sustained relief from her pain so that she can do the things she enjoys.
Voltaren Emulgel 2% provides clinically proven joint pain relief7 in a convenient, twice-daily application, without the worry of adding more oral medications.
Recommend Voltaren Emulgel 2% to your patients for relief of joint pain and inflammation all day long when applied twice daily

Voltaren Emulgel 2%
For patients who are looking for long-lasting relief from joint pain and inflammation, using a topical formulation.
Voltaren 12-hour − up to 12 hours of relief from joint pain7

Joint pain and osteoarthritis
Learn more about osteoarthritis and how it can be managed with physiotherapy.

